Description
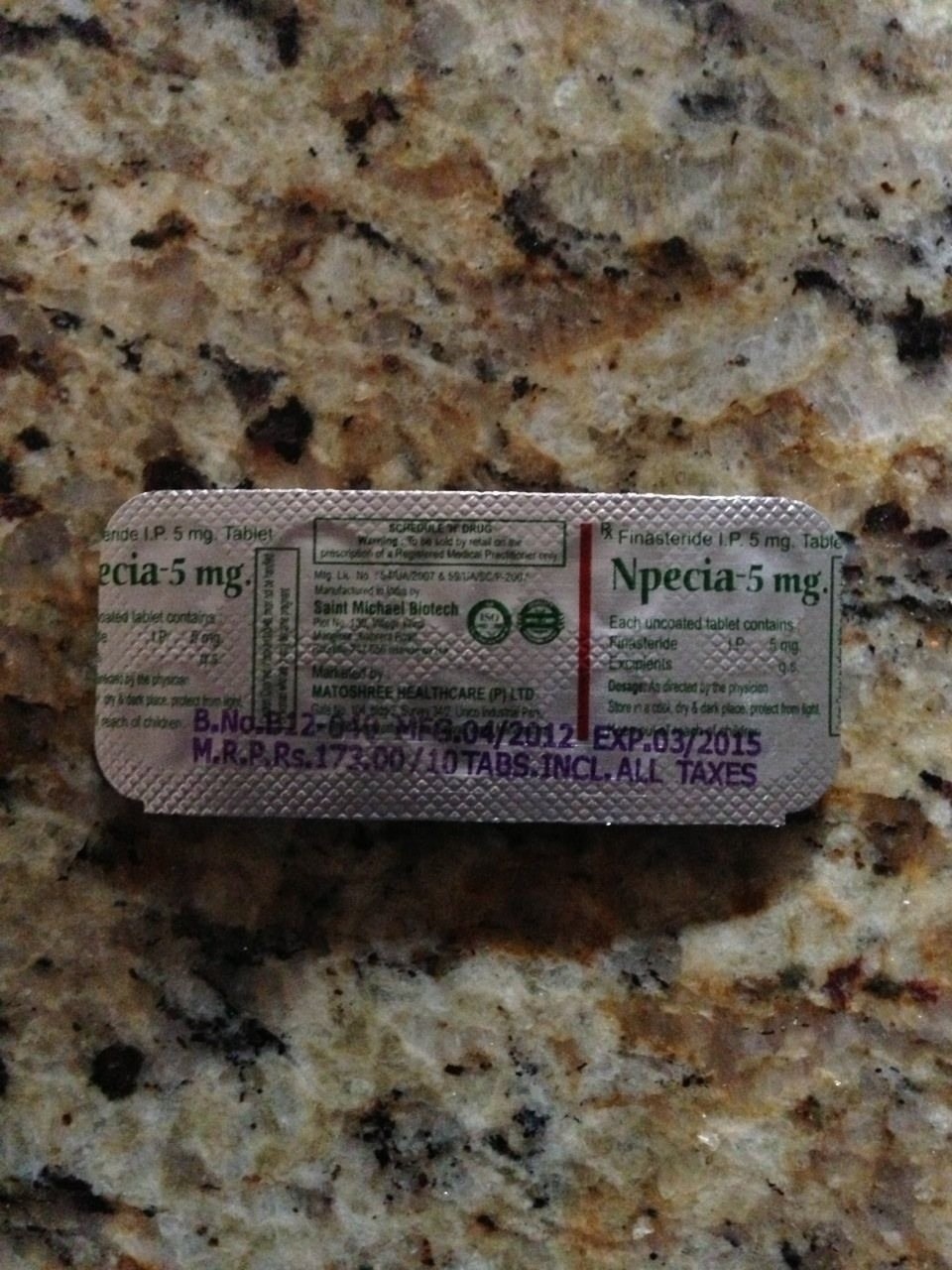
Npecia – synthetic drug, which is intended for the treatment of benign prostatic hyperplasia (BPH) and male pattern baldness. The drug is an inhibitor of 5.alpha.-reductase type II. 5.alpha.-reductase – an enzyme responsible for the conversion of testosterone to dihydrotestosterone (DHT). It is a synthetic 4-azasteroid compound. Good lowers levels of DHT in the blood and prostate tissue. The systematic (IUPAC) name: N- (1,1-dimethylethyl) -3-oxo- (5alpha, 17beta) -4-aza-androst-1-ene-17-carboxylic acid, the formula of C23H36N2O2, molecular weight 372.549 g / mol .
Action
Inhibitor 5-alpha reductase – the enzyme which converts testosterone to dihydrotestosterone, a more active. Reduces the concentration of DHT in the blood and the prostate tissue. It inhibits the stimulatory effect of dihydrotestosterone on the development of benign prostatic hyperplasia.
Side effects of Npecia
Cessation of side effects observed in men who discontinued treatment, and the majority of men who have continued.
In December 2010, Merck added depression to the list of side effects of the drug. In November 1997, the FDA declined to approve Propecia is a drug for the treatment of male pattern baldness.
It positions itself as an international organization of men who suffer from persistent sexual, mental and physical side effects caused by the drug Npecia (Propecia, Proscar and generics), used to treat male pattern baldness.